Abstract
C-phycocyanin (C-PC) is a high-value pigment–protein in Spirulina biomass whose thermolability makes it vulnerable to degradation during post-harvest drying, yet freeze-drying—the gold-standard preservation method—remains prohibitively energy-intensive for large-scale production. This study investigated whether controlled-humidity air drying can preserve C-PC content and its antioxidant functionality in dried biomass comparably to freeze-drying. Wet Spirulina biomass (25 g fresh weight) was dried under nine factorial combinations of temperature (25, 40, and 60°C) and relative humidity (20, 30, and 40%) using a temperature–humidity chamber, with freeze-drying (FD) as the reference. C-PC was extracted, quantified spectrophotometrically along with allophycocyanin and chlorophyll, and its purity index (A620/A280) and antioxidant activity (DPPH radical scavenging, IC50, and ascorbic acid equivalent antioxidant capacity) were determined. Statistical analyses included one-way ANOVA with Tukey's post-hoc test and partial Pearson correlation. Air drying at 25°C yielded C-PC contents of 184.39 - 194.72 mg g-1 DW (98 - 104% of the FD reference, 188.11 mg g-1 DW) with food-grade purity indices of 0.95 - 1.00, whereas drying at 40°C and 60°C reduced C-PC to 76 - 85% and 58 - 63% of the FD value, respectively. Partial correlation analysis identified temperature as the dominant determinant of C-PC loss (Pearson r = -0.90 to -1.00), while relative humidity exerted a secondary, modulatory influence. The 25°C air-dried C-PC retained 86 - 87% of the antioxidant capacity of a commercial C-PC reference standard, approaching the 94% retention achieved by freeze-drying. These findings demonstrate that controlled-humidity air drying at 25°C and 20 - 30% relative humidity effectively preserves both C-PC yield and bioactivity at levels comparable to freeze-drying, offering a potential scalable and low-cost drying alternative for the Spirulina nutraceutical industry
1. Introduction
Spirulina is widely cultivated as a nutrient-dense cyanobacterial biomass and is frequently commercialised as a dried powder to stabilise the product that otherwise contains very high moisture and degrades rapidly after harvest [1, 2]. Among its high-value constituents, C‑phycocyanin (C‑PC) is a dominant blue photosynthetic pigment–protein complex that is both technologically attractive as a natural colorant and biologically relevant as a bioactive ingredient [3, 4]. Robust Spirulina production requires drying, but the conventional methods can damage its valuable ingredients e.g., phycobiliproteins (PBPs). Therefore, finding scalable methods that dry the biomass as well as preserve its nutrient quality is a key industrial challenge for Spirulina-based foods and nutraceuticals [1, 5].
C‑PC belongs to the family of PBPs, which are water‑soluble, chromophore‑containing accessory light‑harvesting proteins in cyanobacteria and some algae [6, 7]. PBPs consist of α and β polypeptide subunits that assemble as αβ heterodimers and higher oligomers (trimers/hexamers) that form the phycobilisome complexes on the thylakoid membrane that broadens light absorption and funnels excitation energy toward the photosystems [2, 6]. In phycobilisomes, energy transfer typically proceeds from phycoerythrin (PE) (when present) to phycocyanin (PC) to allophycocyanin (APC) and then to chlorophyll (Chl) in the photosystems [6]. At the molecular level, C‑PC is covalently attached to linear tetrapyrrole chromophores (phycobilins). In Arthrospira platensis, the chromophore is commonly described as phycocyanobilin (PCB), chemically linked to cysteine residues through thioester bonds, yielding a strong absorbance around 620 nm that is routinely used for quantification and for assessing denaturation/purity via A620/A280 ratios [4, 8]. These chromophore–protein features underpin both the intense blue coloration and the sensitivity of C‑PC to physicochemical stresses relevant to food processing.
C‑PC is abundant in cyanobacteria, e.g., Arthrospira sp., Synechococcus sp., are among the most commercially important sources because they can be produced at scale and yield high protein and pigment contents [4, 9]. Spirulina is repeatedly cited as a rich source, with some reports noting that C‑PC may represent a substantial fraction of dry biomass (e.g., ~20% in one drying-focused study; ~14% of dry weight in another review), although values vary with strain and cultivation conditions [7, 10]. PC is also reported across cyanobacteria and, to a lesser extent, in some red algae and cryptomonads, supporting the broader natural-colorant relevance of PBPs [7, 10]. C‑PC and/or its chromophore PCB have been associated with a wide range of bioactivities, particularly antioxidant and anti‑inflammatory actions [4, 7]. Mechanistically, the chromophore PCB has been implicated as a major driver of some antioxidant effects, including reactive‑species scavenging and modulation of antioxidant defense pathways (e.g., effects linked to heme oxygenase‑1 and inhibition of NADPH oxidase/NOX in preclinical models) [4]. Reviews describing Spirulina PBPs also highlight broader reported activities (e.g., anticancer, immunomodulatory, and hepato‑/neuroprotective effects) and emphasise the relevance of C‑PC as a nutraceutical component beyond its colouring function [11].
Drying is indispensable for microalgal biomass valorisation but is also among the most energy‑intensive and cost‑dominant steps in industrial production chains [5, 12]. From a quality standpoint, multiple studies converge on the conclusion that C‑PC is thermolabile and that drying temperature is a principal driver of losses. For example, in a comparative Spirulina-drying analysis, atmospheric (mild) drying preserved a large fraction of C-PC, whereas employing higher temperatures—particularly above 50°C—caused a sharp decrease in C-PC content [13]. Similarly, work comparing drying/storage modes reported that oven drying can damage heat‑sensitive molecules, while freeze-drying better preserves quality but is slow and costly and may not always maximize every antioxidant metric [9]. Direct comparisons of freeze‑ and hot‑air/oven drying in Spirulina platensis powder illustrate the magnitude of method dependence: freeze-drying (−60°C, 22 h) yielded 27.3% C‑PC with a reported A620/A280 purity of 1.26, whereas oven drying (80°C, 7 h) produced lower C‑PC (with an associated ~35.4% loss relative to freeze drying) with reduced purity (A620/A280 1.16), and ~20% C-PC content loss was reported in spray-dried biomass [10, 14]. Complementary process-focused work also notes that phycobiliproteins begin to thermally degrade above 40-45°C, with rapid denaturation at higher temperatures, underscoring the importance of ambient-temperature or short-exposure strategies [1, 8]. Refractance-window drying is presented as a faster, more energy‑efficient alternative with product quality (colour and nutrient retention) reportedly close to freeze-dried products, but with clear constraints: C-PC was negatively affected by higher temperatures (> 80°C) and prolonged exposure (> 4.5 h) [15]. Likewise, infrared (IR) drying of Spirulina reduced C-PC relative to fresh biomass, but intermittent IR schedules improved retention and shortened drying time compared with continuous conditions, illustrating how dynamic thermal profiles may mitigate degradation [16]. Importantly for air drying, convective-air drying research explicitly treats humidity as a key control variable: process parameters including “temperature, humidity, air velocity” were linked to antioxidant retention, and a two-step temperature strategy was proposed to reduce total drying time while retaining more antioxidants, with major C-PC loss occurring in the falling-rate period [17]. These findings collectively suggest that not only temperature but also the moisture-removal trajectory (and the coupled temperature–moisture history) can shape C‑PC degradation.
Although numerous studies have compared drying technologies for Spirulina and other cyanobacteria and microalgae, the evidence base is fragmented across drying modalities, scales, and endpoints (pigment content, purity, color metrics, or selected biological assays), and controlled humidity is rarely treated as a primary design variable in Spirulina C‑PC preservation studies. Yet humidity is explicitly recognised as a key parameter affecting retention of biological activities in convective-air drying, and microstructure changes that alter oxygen exposure during drying (e.g., porous freeze-dried structures) can plausibly modify oxidation-driven losses [13, 17]. At the same time, industrial decision-making must address energy and cost: freeze-drying is widely recognized for quality preservation but is expensive and energy demanding, while spray drying can also incur high energetic costs and may expose pigments to high temperatures [9, 12]. These constraints motivate the development of air‑drying approaches that are more energy-efficient than spray-drying yet more protective than conventional hot-air drying, particularly for heat- and oxidation-sensitive compounds such as C‑PC [5, 15].
The present work, therefore, evaluates controlled-humidity air drying as a practical lever to improve C‑PC retention in dried Arthrospira (Spirulina) platensis biomass. Building on prior evidence that (i) C‑PC degradation is highly temperature dependent and accelerates above ~45-50°C [8, 13], (ii) drying-stage dynamics influence losses (falling-rate sensitivity) [18], and (iii) humidity is a meaningful process variable linked to biological activity retention in convective drying [18, 19], this study tests whether adjusting relative humidity (RH) at different drying temperatures can shift the drying dynamics to better preserve pigment and function. Specifically, this study quantifies (i) retained C‑PC content in the dried biomass, (ii) drying rate/speed as a process-performance metric, and (iii) DPPH radical scavenging activity as functional evidence for retained biological activity of the resultant C-PC-associated antioxidant capacity. By jointly analysing C‑PC retention, drying kinetics, and antioxidant activity under controlled RH–temperature combinations, this work aims to clarify whether humidity control can provide a scalable, energy‑conscious alternative to highly energy‑intensive preservation routes while maintaining the quality attributes that motivate Spirulina’s use as a source natural colorant and active biological ingredients.
2. Materials and Methods
2.1. Chemicals and reagents
Anhydrous sodium hydrogen phosphate (013437), disodium hydrogen phosphate heptahydrate (3803), and sodium hydroxide pellets (BP359) were procured from Thermo Fisher Scientific (USA). L-ascorbic acid (47863) was obtained from Supelco (USA). 2,2-Diphenyl-1-picrylhydrazyl (D9132) and lyophilised C-phycocyanin reference standard (P2172) were purchased from Sigma-Aldrich (Germany). The lyophilised C-PC powder served as the reference standard for comparative analyses.
2.2. Spirulina biomass
The cyanobacterium Arthrospira (Spirulina) platensis was procured from the Spirulina Pilot Plant, Applied Botany Laboratory, BCSIR Laboratories, Dhaka, Bangladesh. This indigenous strain is routinely cultivated in open raceway ponds under natural diffuse sunlight within the facility's greenhouse enclosure [20]. Biomass production uses Bangladesh Medium 1 (Bd1), a culture medium specifically formulated to sustain consistent year-round growth of this locally adapted cyanobacterium [20, 21]. The complete compositional details of the Bd1 medium are provided in Supplementary Table S1.
Raw biomass was harvested during the late-exponential growth phase (OD560 ≈ 1.35) and subjected to preliminary decontamination. The harvested biomass was thoroughly rinsed with double-distilled water to eliminate residual culture medium and loosely adhering contaminants, followed by three successive washing cycles involving centrifugation (5,000 × g, 4°C, 10 min) with fresh water each cycle. Under these centrifugal conditions, denser particulate matter and non-buoyant contaminants sedimented, whereas Spirulina filaments preferentially accumulated near the surface due to the presence of gas vesicles, thereby facilitating effective physical separation [22]. The recovered biomass was subsequently treated with 0.1 M sodium bicarbonate (NaHCO3, pH 9.0) for 30 min at ambient temperature, utilising its alkaline stress tolerance to reduce residual microbial contaminants [23]. A final centrifugation step was performed to remove remaining suspended impurities. The buoyant biomass fraction was collected, strained through sterile mesh, and repeatedly washed with double-distilled water to eliminate residual bicarbonate. The resultant decontaminated biomass slurry was used immediately for drying experiments.
2.3. Biomass drying techniques
Borosilicate Petri dishes (120 mm diameter, 15 mm depth) were prepared containing 25 g fresh weight of decontaminated Spirulina slurry, uniformly spread to an approximate thickness of 5 mm. Controlled air-drying experiments were conducted inside a benchtop temperature-humidity chamber (CGG-80, MRC, Germany) employing factorial combinations of three temperatures and three relative humidity levels: 25°C-20% RH (T1H1), 25°C-30% RH (T1H2), 25°C-40% RH (T1H3), 40°C-20% RH (T2H1), 40°C-30% RH (T2H2), 40°C-40% RH (T2H3), 60°C-20% RH (T3H1), 60°C-30% RH (T3H2), and 60°C-40% RH (T3H3).
Freeze-drying served as the reference method (FD) against which all air-drying outcomes were evaluated, given its established efficacy in preserving thermolabile phycobiliproteins [24, 25]. Biomass-containing plates were pre-frozen at −20°C for 2 h and subsequently dried in a vacuum freeze dryer (BK-FD18PT, BIOBASE, China). Primary drying was conducted at a condenser temperature of −80°C under vacuum (<5 Pa) for 6 h. Secondary drying proceeded for an additional 2 h with gradual elevation of the shelf temperature to 25°C while maintaining vacuum conditions. All drying experiments were performed in three independent replicates.
All air-drying processes were continued until stable dry mass was achieved for each sample, with corresponding drying durations recorded. Residual moisture content was subsequently determined using a high-performance moisture analyser (WBA-110M, Witeg, Germany) and expressed as a percentage of the corresponding sample weight (wet basis). Absolute dry weight (g DW) of each dried biomass was calculated by subtracting moisture content from the respective biomass weight. Following drying, all samples were pulverised, sieved through an 80-mesh screen, and stored in airtight, light-protected containers at 4°C until further analysis.
2.4. C-PC extraction method
Total C-PC content in the dried biomass was quantified spectrophotometrically from crude extracts obtained through exhaustive freeze–thaw extraction [26]. Briefly, 0.1 g dry weight of each powder was suspended in 10 mL of cold extraction buffer (50 mM sodium phosphate, pH 6.8). Suspensions were vortexed thoroughly and subjected to sequential freeze–thaw cycles (−20°C for 4 h, followed by thawing at 4°C). After centrifugation (10,000 × g, 4°C, 10 min), the supernatant was collected, and the pellet was resuspended in fresh buffer; this process was repeated until the supernatant became colourless. All supernatants were pooled, filtered through a 0.22 μm polyethersulfone (PES) syringe filters, and stored at 4°C in light-proof containers. Absorbance spectra were recorded using a double-beam UV-Vis spectrophotometer (UV-1800, Shimadzu, Japan) for C-PC quantification. C-PC content was expressed as estimated yield in milligrams of C-PC per gram dry weight powder (mg g-1 DW).
2.5. Spectroscopic analysis and quantification
UV-Vis spectrophotometry was employed for C-PC quantification in all liquid extracts. A 250 µg mL-1 solution of the C-PC reference standard was concurrently scanned alongside all the prepared C-PC extracts for comparative spectral analysis. Absorbance spectra were recorded across the 250–800 nm wavelength range using double-distilled water as blank, unless otherwise specified. All spectra were recorded in triplicate, baseline-corrected at 800 nm, normalised at 620 nm, and presented as mean spectra for each sample.
C-PC and APC exhibit characteristic absorbance maxima (λmax) at 620 nm and 652 nm, respectively, in aqueous solution. C-PC concentration, purity index, and yield were calculated using equations originally described by Bennett and Bogorad (1973), incorporating corrections for chlorophyll interference at 620 nm (Eq. 1) and 652 nm (Eq. 2) as proposed by Lauceri et al. (2017) [27, 28]:
where A and A* represent raw and corrected absorbance, respectively; A620, A652, A675, and A280 correspond to absorbance of C-PC, APC, Chl, and residual proteins, respectively.
C-PC and APC concentrations were determined using Eq. 3 and Eq. 4, respectively:
where DF denotes the dilution factor.
Residual Chl concentration was estimated using Eq. 5, developed by Porra (2002) for total chlorophyll determination in aqueous systems [29]:
C-PC purity index was calculated according to Eq. 6:
Estimated C-PC yield was expressed on a dry weight basis using Eq. 7:
Chl separation factor was was calculated by the ratio of A*620 to A675, according to Eq. 8:
2.6. DPPH scavenging activity
Antioxidant activity was assessed using the 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay, based on the method described by Blois (1958), with modifications adopted from Choi & Lee (2018) [30, 31]. Aqueous C-PC extracts were subsequently frozen and lyophilised following the procedure described in Section 2.3. Lyophilised C-PC samples and the reference standard were dissolved in 80% (v/v) methanol at concentrations ranging from 100 to 1500 µg mL-1 and tested against 0.1 mM DPPH prepared in the same solvent. Ascorbic acid (AA) (1–10 µg mL-1) served as a reference antioxidant. Control reactions contained 0.1 mM DPPH without any antioxidant. Reaction mixtures were incubated in darkness at ambient temperature for 30 min, after which absorbance was measured at 517 nm against the solvent blank. The scavenging rate was calculated using Eq. 9:
where AS, AC, and AB represent the absorbance of the sample, control, and blank, respectively. The concentration required for 50% radical scavenging activity (IC50) was determined from concentration versus percentage inhibition plots using linear regression analysis. Results represent the means of triplicate measurements.
2.7. Statistical analysis
All data are presented as mean ± standard deviation of triplicate measurements. Statistical analyses were performed using IBM-SPSS Statistics for Windows, Version 30.0 (IBM Corp., 2024) [32]. Quantitative differences among experimental groups were evaluated using one-way analysis of variance (ANOVA), followed by Tukey's post-hoc pairwise comparison test for multiple comparisons. Partial correlation was estimated between independent (temperature and relative humidity) and dependent (C-PC yield, purity, and Chl separation factor) variables using Pearson’s correlation coefficient (r). Statistical significance was established at a 95% confidence level (p < 0.05).
3. Results
3.1. Physical characteristics of the dried biomass
The drying conditions employed in this study produced distinctly different physical outcomes in terms of drying duration, dry biomass yield, residual moisture content, and absolute dry mass, as summarised in Table 1. All air‑drying treatments were continued until stable mass was attained, and the required drying periods varied widely depending on the temperature–humidity combination used.
Among the air‑dried preparations, drying temperature was the primary determinant of drying speed. At 60°C, the biomass reached a stable mass in 10.0 - 12.67 h across the three RH levels tested, which was 1.6‑ to 1.8‑fold faster than at 25°C, where 16.33 - 22.67 h was required (Table 1). The 40°C group occupied an intermediate range of 14.17 - 18.67 h. Freeze‑drying (FD), serving as the reference technique, required a total processing period of 8.0 h.
At 25°C the apparent dry mass rose from 8.01 ± 0.33 g at 20% RH to 10.76 ± 0.19 g at 40% RH, reflecting the higher residual moisture content retained at elevated humidity. The freeze-dried biomass retained the lowest moisture at 7.46 ± 0.11%, consistent with the efficacy of sublimation under vacuum conditions [24, 25]. Correspondingly, the residual moisture content scaled with the ambient RH in a near proportional manner: at 20% RH the endpoint moistures were 17.73%, 15.18%, and 13.93% for 25, 40, and 60°C, respectively, whereas at 40% RH they rose to 40.96%, 39.91%, and 37.53% for the same temperatures (Table 1).
The 25°C group yielded the highest absolute dry masses (6.35 - 6.59 g DW from 25 g fresh weight), which were statistically comparable to one another and were, notably, higher than the FD reference (6.03 ± 0.23 g DW; p < 0.05). This observation suggests that gentler convective drying at ambient temperature minimised thermally induced losses of volatile organic matter that may occur even during the secondary drying phase of freeze drying. In contrast, absolute dry mass declined progressively at higher drying temperatures: the 40°C group yielded 5.34 - 5.94 g DW, and the 60°C group yielded 5.19 - 5.47 g DW. The lowest absolute dry mass was recorded for T3H3 (5.19 ± 0.09 g DW), which was significantly lower than all 25°C treatments and the FD reference (p < 0.05).
3.2. C-PC contents in the dried biomass
UV Vis absorbance spectra of C-PC extracts obtained from all dried preparations (Figure 1) displayed the characteristic absorption maximum at approximately 620 nm, confirming the presence of C-PC across all drying conditions. Baseline corrected spectra (Figure 1a) showed that the absolute peak height at 620 nm followed a clear hierarchy: extracts from 25°C dried biomass exhibited the strongest absorbance, marginally matching the FD reference, whereas 40°C preparations showed intermediate absorbance and 60°C preparations exhibited the weakest 620 nm peaks. The normalised spectra (Figure 1b), adjusted to unit absorbance at 620 nm, facilitated comparison of spectral shape and secondary features. A secondary absorption shoulder at approximately 652 nm, attributable to APC, and a minor absorption band near 675 nm from residual Chl were discernible in all spectra.
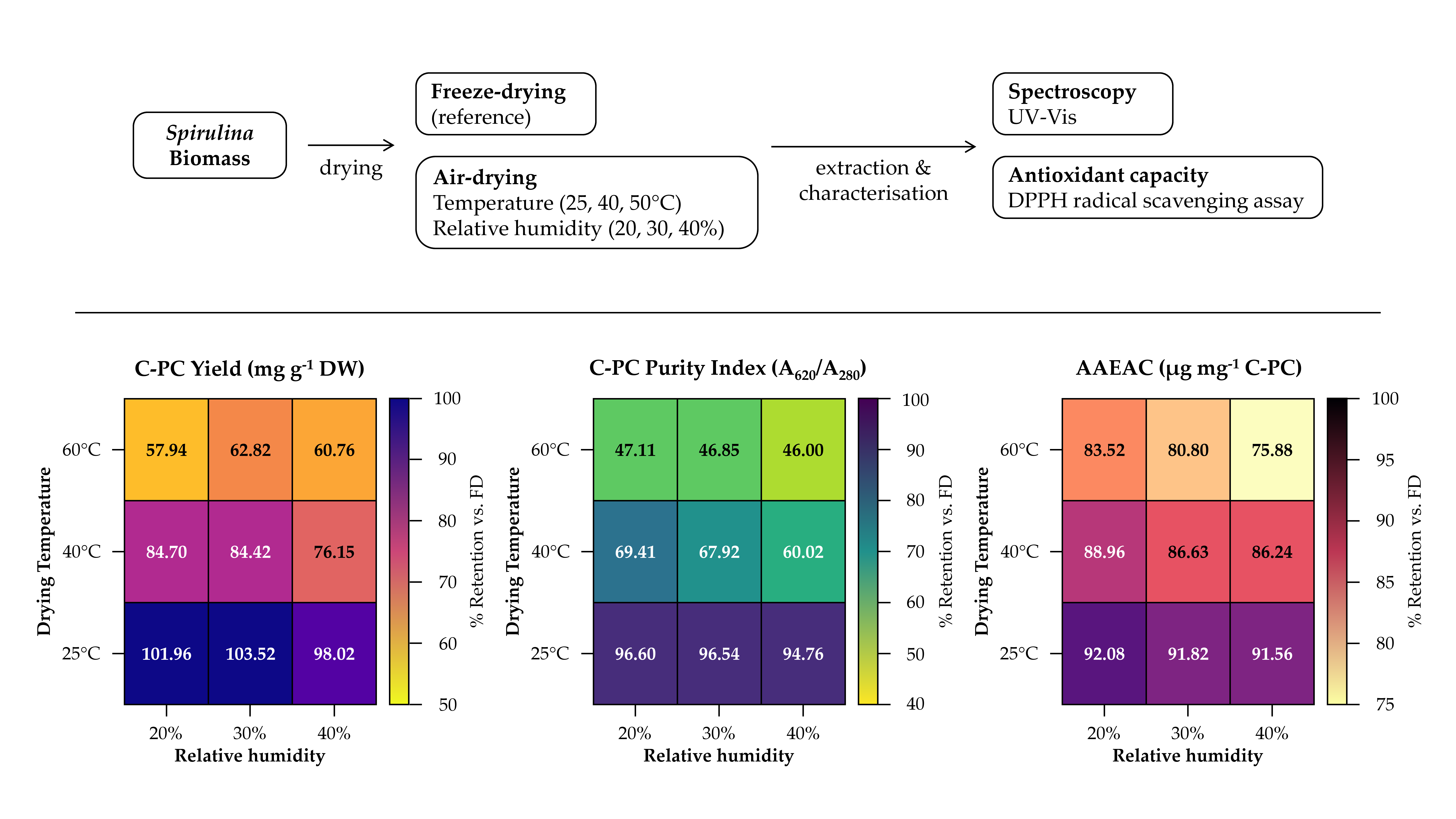
Quantitative estimations of C-PC yield, purity index (PI), and Chl separation factor (SF) are presented in Table 2 and Figure 2, with supporting concentration data for C PC, APC, and Chl provided in Supplementary Table S2. The C-PC yield ranged from 108.98 ± 2.75 to 194.72 ± 4.08 mg g-1 DW across all drying treatments. The highest C-PC yield was obtained from T1H2 (25°C, 30% RH), at 194.72 ± 4.08 mg g-1 DW, which was not statistically different from FD (188.11 ± 0.49 mg g-1 DW) or T1H1 (191.79 ± 3.71 mg g-1 DW). Drying at 40°C resulted in a significant decrease in C-PC yield relative to the 25°C group and FD. The 40°C preparations yielded 143.25 - 159.33 mg g-1 DW, corresponding to 76.2 - 84.7% retention relative to FD (Table 2). Within this group, increasing RH from 20% to 40% was associated with a further decline from 159.33 ± 2.77 (T2H1) to 143.25 ± 2.42 mg g-1 DW (T2H3), with T2H3 being significantly lower than both T2H1 and T2H2 (p < 0.05). Drying at 60°C caused the most severe C-PC losses: yields ranged from 108.98 ± 2.75 (T3H1) to 118.16 ± 0.96 mg g-1 DW (T3H2), corresponding to only 57.9 - 62.8% retention relative to FD.
The CPC PI (A*620/A280) mirrored the yield trend (Table 2; Figure 2a). The FD and all 25°C preparations shared the highest purity values (0.95 - 1.00), indicating that the extracted CPC was of comparable compositional quality to the reference standard. Purity fell sharply at 40°C (0.60 - 0.70) and further at 60°C (0.46 - 0.47). The PI declined approximately 30% from the 25°C group mean (0.96) to the 40°C group mean (0.66), and by a further 29% to the 60°C group mean (0.47), reflecting progressive protein denaturation and coextraction of nonCPC proteins at higher temperatures.
The SFChl (A*620/A675) provided additional insight into CPC integrity and chlorophyll contamination (Table 2; Figure 2b). FD (11.35 ± 0.03) and T1H1 (11.20 ± 0.05) exhibited the highest and statistically equivalent SFChl, indicating minimal chlorophyll interference in the 620 nm absorbance measurement and wellpreserved CPC chromophore structure. Within the 25°C group, T1H2 and T1H3 showed lower but still relatively high Chl separation factors (8.41 and 8.77, respectively), suggesting a modest increase in Chl coextraction with rising RH even at low temperature. Interestingly, T2H1 (40°C, 20% RH) exhibited a Chl separation factor (9.13) that was numerically intermediate between the two extremes of the 25°C subgroup—higher than T1H2 and T1H3 but lower than FD and T1H1. However, the SFChl declined dramatically within the 40°C group as RH increased (9.13 → 6.61 → 3.05 for T2H1 → T2H2 → T2H3), and was severely reduced across the entire 60°C group (1.90 - 2.33). Each 60°C preparation had a significantly different Chl separation factor from the others (T3H1 > T3H2 > T3H3; p < 0.05), highlighting that even at high temperature, RH continues to exert a measurable negative effect on spectral purity.
Concurrent APC and Chl concentration data (Supplementary Table S2; Figure 2b) revealed complementary trends. CPC concentration in crude extracts decreased progressively from the 25°C group (2.31–2.43 mg mL-1) through 40°C (1.79 - 1.99 mg mL-1) to 60°C (1.36 - 1.48 mg mL-1), paralleling the yield data. Residual Chl concentrations remained relatively uniform across all preparations (0.12 - 0.15 mg mL-1).
3. Correlation between the analysed variables
Partial correlation analysis using Pearson's correlation coefficient revealed contrasting strengths of association between the two independent drying variables (temperature and RH) and the three C‑PC quality metrics (Table 3). Temperature, when evaluated at constant RH, exhibited uniformly strong and negative correlations with C‑PC yield (r = -0.98 to -1.00), PI (r = -0.95 to -0.99), and SFChl (r = -0.90 to -0.99) across all three RH levels. Among the RH strata, the strongest temperature correlations were observed at 20% and 30% RH, where all three metrics showed |r| ≥ 0.98, whereas at 40% RH the correlations were slightly attenuated (|r| = 0.90 - 0.98), suggesting marginally greater variability in C‑PC outcomes when drying at higher humidity. In contrast, the effect of RH on C‑PC metrics, when evaluated at constant temperature, was weaker and more variable. At 25°C, RH showed moderate negative correlations with C‑PC yield (r = - 0.58), purity (r = -0.80), and Chl separation factor (r = -0.87). At 40°C, the corresponding correlations were similar for yield and purity (r = -0.53 and -0.81, respectively) but stronger for Chl separation factor (r = -0.96).
3.4. Antioxidant activities of the resultant C-PCs
The DPPH radical scavenging activities of C-PC preparations from all drying treatments, along with the lyophilised C-PC reference standard and ascorbic acid (AA), are summarised in Table 4, with the corresponding concentration–radical scavenging activity regression plots presented in Supplementary Figure S1. All C-PC preparations exhibited dose‑dependent DPPH scavenging activity with strong linearity (R² = 0.991 - 0.998), confirming that the extracted C-PC retained measurable radical scavenging capacity regardless of the drying method employed. The lyophilised C-PC reference standard exhibited the highest antioxidant potency among all C‑PC preparations, with an IC50 of 680.55 µg mL-1 and an AAEAC of 7.71 µg AAE mg-1 C‑PC.
All three 25°C air‑dried preparations (T1H1, T1H2, T1H3) formed a statistically homogeneous group for both IC50 (786.35 - 790.85 µg mL-1) and AAEAC (6.63 - 6.67 µg AAE mg-1 C-PC), retaining 86.0 - 86.5% of the reference standard's antioxidant capacity. At 40°C, antioxidant activity was moderately reduced, with IC50 values of 813.96 - 839.59 µg mL-1 and AAEAC values of 6.25 - 6.44 µg AAE mg-1 C-PC, retaining 81.1 - 83.5% of the reference standard activity. The 60°C-dried preparations exhibited the weakest antioxidant activities among all C‑PC preparations, with IC50 values rising from 866.91 (T3H1) to 954.25 µg mL-1 (T3H3) and AAEAC declining from 6.05 to 5.50 µg AAE mg-1 C-PC—corresponding to only 71.3–78.5% of the reference standard. The highest IC50 (lowest antioxidant potency) was recorded for T3H3 (60°C, 40% RH; IC50 = 954.25 µg mL-1), which was significantly different from all other preparations except T3H2 for IC50.
4. Discussion
Within each temperature group, increasing RH consistently prolonged the drying period: at 25°C, raising the RH from 20% to 40% extended the drying time from 16.33 ± 0.29 h to 22.67 ± 0.56 h (p < 0.05); analogous trends were observed at 40°C (14.17 → 18.67 h) and 60°C (10.00 → 12.67 h). This behaviour is expected because a higher ambient vapour pressure reduces the driving force for evaporation, slowing moisture removal and requiring longer residence times to reach equilibrium [5, 15, 18]. FD was the shortest overall treatment; however, this comparison should be considered alongside the substantially greater energy intensity and equipment complexity of the freeze‑drying process [1, 24, 25]. The apparent dry biomass yield (i.e., mass remaining after drying from 25 g fresh weight) increased markedly with ambient RH at each temperature level, because the dried product equilibrated with the ambient moisture rather than achieving absolute dryness. When adjusted for residual moisture, the absolute dry mass (g DW) revealed an important pattern related to volatile and soluble component losses during drying. The overall pattern indicates that drying at elevated temperatures promotes greater loss of total dry matter, likely through enhanced volatilisation and/or oxidative degradation of organic constituents [1, 5]. This dry matter loss at higher temperatures has practical implications, as it reduces not only biomass recovery but also the total pool of extractable bioactive compounds including C-PC. Additionally, low-humidity limits microbial proliferation, and accelerate the ambient-temperature drying as reported by Ughetti et al. (2024) and Alp & Bulantekin (2021) [18, 33]. At each RH level, higher temperatures produced slightly lower residual moisture, consistent with the greater thermodynamic driving force for desorption at elevated temperature.
The relative prominence of the 652 nm (APC) and 675 nm (Chl) shoulders increased progressively in spectra of the highertemperature preparations, indicating a compositional shift in the extracted PBPs and greater Chl contamination at elevated drying temperatures. An absorbance band near 280 nm, attributable to aromatic amino acid residues and total protein content, was observed in all extracts, and the proteintopigment spectral ratio (280 nm vs. 620 nm) was evidently altered in the hightemperature preparations, reflecting reduced purity of CPC in those extracts. All T1 preparations yielded CPC values of 184.39 - 194.72 mg g-1 DW, representing 98.0 - 103.5% of the FD reference—demonstrating that ambienttemperature controlledhumidity air drying at 25°C preserves CPC content as effectively as freezedrying. This finding is consistent with prior reports that CPC remains stable at temperatures below ~45°C [8, 9, 13] and that freezedrying, while a gold standard, does not necessarily outperform wellcontrolled ambienttemperature convective drying for PBP retention [2, 14]. Güroy et al. (2017) reported 35.4% CPC loss during convective drying at 80°C relative to freezedrying [10]; in comparison, the present 25°C airdrying conditions avoided any statistically significant loss relative to FD, underscoring the critical importance of maintaining subthreshold temperatures. The humiditydependent decline within the 40°C group is noteworthy because 40°C approaches but does not exceed the commonly reported thermal denaturation onset (45-50°C) [8, 9]; the additional stress from prolonged drying at higher RH (18.67 h at 40% RH vs. 14.17 h at 20% RH) likely amplified timedependent degradation. All three 60°C treatments indicated no significant variation among them despite their different RH levels. This plateauing of losses at 60°C suggests that the thermal denaturation of CPC at this temperature is sufficiently rapid and complete that modulating RH provides little additional protective benefit—consistent with the dramatic reduction in halflife of CPC reported at temperatures above 50°C [8, 9].
A PI near or above 0.7 is generally considered acceptable for foodgrade CPC [7], and the 25°C preparations comfortably exceeded this threshold. The 60°C purity values (≤ 0.47) fall below the foodgrade threshold, indicating that the CPC extracted from 60°Cdried biomass would require further purification for commercial applications [7, 14]. Results of SFChl suggest that rapid, lowhumidity drying at moderately elevated temperature can still maintain reasonable CPC chromophore integrity and consistent with substantial loss of CPC chromophore integrity and/or enhanced release of Chl from the biomass matrix under harsher thermal conditions. The 60°C group exhibited a mean SFChl of only 2.14 ± 0.22, representing approximately 19% of the FD value, indicating that the CPC extract obtained from 60°Cdried biomass is dominated by chlorophyll interference and degraded CPC chromophores. The sharp decline in the SFChl at 60°C indicates that the relative contribution of Chl to the 620 nm absorption region becomes increasingly significant as CPC degrades—a point of practical importance for spectrophotometric quantification and quality assessment [27, 37]. The concomitant reduction in the CPCtoAPC concentration ratio from approximately 6.0 (FD and 25°C group) to approximately 3.0 (60°C group) is consistent with the known spectral behaviour of partially degraded or dissociated CPC: thermal denaturation promotes disassembly of hexameric and trimeric CPC into lower oligomeric forms whose spectral properties shift toward the 652 nm region characteristic of APC [6, 8, 9].
The near‑perfect negative correlations (|r| ≥ 0.90 in all 9 temperature‑controlled comparisons) confirm that drying temperature is the dominant and most consistent driver of C‑PC degradation, in agreement with the well‑established thermal sensitivity of PBPs that begin to denature appreciably above approximately 45–50 °C [8, 9, 13]. The Chl separation factor was the most sensitive metric to RH changes at every temperature level, with near‑perfect correlations at both 40°C (r = -0.96) and 60°C (r = -1.00), indicating that increasing RH exacerbates Chl contamination of the C‑PC extract. This likely occurs because higher RH prolongs the drying period, thereby extending the biomass exposure to conditions that promote Chl release from degrading thylakoid membranes and/or C‑PC chromophore deterioration [18, 19]. An anomalous finding was observed at 60°C, where the correlation between RH and C‑PC yield was weakly positive (r = +0.53), in contrast to the negative correlations seen at 25°C (r = -0.58) and 40°C (r = -0.53). This reversal may reflect a moisture‑buffering mechanism: at 60°C, where thermal stress is severe and the drying rate is rapid, higher RH may slow the rate of water loss during the falling‑rate period, providing evaporative cooling that partially moderates product temperature rise and thereby partially offsets thermally driven C‑PC denaturation [5, 18]. However, this modest positive effect on C-PC yield did not translate into improved PI or SFChl, both of which remained strongly negatively correlated with RH at 60°C (r = -0.93 and -1.00, respectively), indicating that the overall quality of C‑PC continued to deteriorate with rising humidity at the highest temperature. This dissociation between yield and quality metrics at 60°C suggests that while the total extractable C‑PC mass may be marginally preserved by humidity buffering, the structural integrity of the pigment-protein complex is nonetheless compromised.
Ascorbic acid, included as a positive reference antioxidant, displayed an IC50 of 5.24 µg mL-1—approximately 130‑fold more potent than C‑PC on a mass basis. This large difference is expected given that C‑PC is a high‑molecular‑weight protein–pigment complex (~36 kDa per αβ monomer) whose radical scavenging activity derives from its phycocyanobilin chromophore and thioether‑linked cysteine residues, whereas ascorbic acid is a small‑molecule reductant that operates at far lower stoichiometric concentrations [33, 34, 35]. Among the experimentally produced C‑PCs, FD C‑PC exhibited the strongest antioxidant activity, with an IC50 of 724.06 µg mL-1 and AAEAC of 7.24 µg AAE mg-1 C-PC—corresponding to 93.9% of the reference standard's AAEAC. This was significantly different from the reference standard (p < 0.05) and from all air‑dried preparations, but the modest IC50 increase of only 6.4% relative to the reference standard indicates excellent preservation of antioxidant functionality by freeze‑drying, consistent with the minimal C‑PC degradation observed in the yield and purity data for this treatment. The moderate DPPH scavenging potency of C‑PC observed here (IC50 in the 680 - 955 µg mL-1 range) is consistent with published reports for crude to food‑grade C‑PC, where IC50 values vary widely depending on purity, extraction conditions, and assay methodology [36, 38]. Relative to FD, the 25°C group showed IC50 values only 8.6 - 9.2% higher, demonstrating that ambient‑temperature controlled‑humidity air drying preserves the antioxidant functionality of C‑PC nearly as effectively as freeze‑drying. Importantly, there were no significant differences among the three RH levels at 25°C for either antioxidant metric, indicating that humidity variation within the 20-40% range does not measurably affect C‑PC bioactivity when the drying temperature remains below the thermal denaturation threshold. Within the 40°C group, a non‑significant trend toward lower activity at higher RH was observed, consistent with the C‑PC yield and purity data showing progressive quality decline with increasing humidity at this temperature. Within the 60°C group, increasing RH progressively diminished antioxidant activity—the IC50 rose by 10.1% from T3H1 to T3H3—consistent with the compounded effect of thermal and oxidative stress during prolonged drying at elevated temperature and humidity. The declining regression slopes across drying severity (from 0.051 for the reference standard to 0.040 for T3H3) further reflect the progressive loss of intrinsic radical scavenging capacity per unit mass of C‑PC, attributable to chromophore degradation, protein unfolding, and associated loss of the chromophore microenvironment that stabilises the phycocyanobilin radical scavenging function [35, 36].
5. Conclusion
This study demonstrates that controlled-humidity air drying at ambient temperature is an effective and practical strategy for preserving C-PC content and its associated antioxidant functionality in dried Arthrospira (Spirulina) platensis biomass. The finding that a simple convective drying process can match the gold-standard freeze-drying technique for phycobiliprotein preservation—provided the temperature is maintained below the thermal denaturation onset of C-PC—is of considerable significance for industry-scale Spirulina processing, where freeze-drying remains prohibitively energy-intensive and capital-demanding for large-scale operations. From a practical standpoint, the controlled-humidity approach offers a scalable, low-cost alternative that requires only conventional air-circulation equipment fitted with humidity regulation, making it accessible to small- and medium-scale producers, particularly in tropical and subtropical regions where Spirulina cultivation is widespread. The partial correlation analysis confirmed that temperature is the dominant determinant of C-PC degradation, while humidity plays a secondary, modulatory role most evident in the chlorophyll separation factor. This hierarchy provides clear process-design guidance: maintaining the lowest feasible drying temperature is paramount, and humidity should be kept at low-to-moderate levels (20 - 30%) to minimise drying duration and chlorophyll co-extraction without compromising C-PC yield.
Nevertheless, this work has several limitations that should be acknowledged. The study employed a single Spirulina strain and a fixed initial biomass quantity, so the generalisability of the optimal conditions to other strains, biomass geometries (e.g., thin-film vs. pellet), or larger batch sizes remains to be established. Drying kinetics were characterised by gross average drying periods rather than continuous moisture-loss profiles, which limits the resolution of mechanistic insights into constant-rate versus falling-rate drying behaviour. The antioxidant assessment relied solely on the DPPH radical scavenging assay; complementary assays (e.g., ABTS, ORAC, FRAP) and in vitro or in vivo bioactivity models would provide a more comprehensive evaluation of functional preservation. Furthermore, long-term storage stability of C-PC in the air-dried biomass was not evaluated, and the economic and energy-consumption advantages of controlled-humidity drying over freeze-drying, although implied, were not quantitatively benchmarked. Future studies should therefore investigate the drying kinetics in greater mechanistic detail using continuous moisture-monitoring systems, extend the assessment to multiple Spirulina strains and scale-up configurations, evaluate storage stability under various packaging and environmental conditions, incorporate broader panels of antioxidant and bioactivity assays, and conduct techno-economic analyses comparing the energy footprint and production costs of controlled-humidity air drying against freeze-drying and other emerging low-temperature techniques such as refractance window drying. Such study would further validate and strengthen the commercial viability of this approach and advance the development of quality-preserving, cost-effective drying protocols for the Spirulina nutraceutical industry.
Supplementary Materials: Supplementary information is available online at https://www.scipublications.com/supplementary/6285/Supplementary materials.docx
Author Contributions: Conceptualization, A.S.B. and J.L.M.; Methodology, A.S.B.; Software, A.S.B.; Validation, A.S.B.; Formal analysis, A.S.B. and N.N.H.; Investigation, A.S.B. and N.N.H.; Resources, N.N.H. and C.K.R.; Data curation, A.S.B.; Writing—original draft preparation, A.S.B.; Writing—review and editing, N.N.H., C.K.R. and J.L.M.; Visualization, A.S.B.; Supervision, N.N.H. and J.L.M.; Project administration, J.L.M.
All authors have read and agreed to the published version of the manuscript.
Funding: This research received no external funding
Data Availability Statement: The authors declare that the data supporting the findings of this study are available within the paper and its Supplementary Information files. Raw data files are available from the corresponding author upon reasonable request.
Acknowledgments: The authors would like to express their sincere gratitude to the Bangladesh Council of Scientific and Industrial Research (BCSIR), Ministry of Science and Technology (MoST), Govt. Republic of Bangladesh [Ref. No. 39.02.0000.011.14.169.2023/877; dated 17.09.2023], for supporting this study.
Conflicts of Interest: The authors declare no conflict of interest.
References
- Demarco, M., De Moraes, J. O., Ferrari, M. C., De Farias Neves, F., Laurindo, J. B., & Tribuzi, G. (2021). Production of Spirulina (Arthrospira platensis) powder by innovative and traditional drying techniques. Journal of Food Process Engineering, 45(1). https://doi.org/10.1111/jfpe.13919[CrossRef]
- Seghiri, R., Legrand, J., Hsissou, R., & Essamri, A. (2020b). Comparative study of the impact of conventional and unconventional drying processes on phycobiliproteins from Arthrospira platensis. Algal Research, 53, 102165. https://doi.org/10.1016/j.algal.2020.102165[CrossRef]
- Nouri, E., Abbasi, H., & Rahimi, E. (2018). Effects of processing on stability of water- and fat-soluble vitamins, pigments (C-phycocyanin, carotenoids, chlorophylls) and colour characteristics of Spirulina platensis. Quality Assurance and Safety of Crops & Foods, 10(4), 335–349. https://doi.org/10.3920/qas2018.1304[CrossRef]
- Citi, V., Torre, S., Flori, L., Usai, L., Aktay, N., Dunford, N. T., Lutzu, G. A., & Nieri, P. (2024). Nutraceutical Features of the Phycobiliprotein C-Phycocyanin: Evidence from Arthrospira platensis (Spirulina). Nutrients, 16(11), 1752. https://doi.org/10.3390/nu16111752[CrossRef] [PubMed]
- Pastor, R. L., Pinna-Hernández, M. G., Molina, J. a. S., López, J. L. C., Rubio, M. I. M., & Fernández, F. G. A. (2026). Optimizing the operational conditions for microalgae biomass drying using tray dryers. Scientific Reports, 16(1), 4515. https://doi.org/10.1038/s41598-025-34616-w[CrossRef] [PubMed]
- Patel, S. N., Sonani, R. R., Roy, D., Singh, N. K., Subudhi, S., Pabbi, S., & Madamwar, D. (2022). Exploring the structural aspects and therapeutic perspectives of cyanobacterial phycobiliproteins. 3 Biotech, 12(9), 224. https://doi.org/10.1007/s13205-022-03284-2[CrossRef] [PubMed]
- Soni, A., Dubey, M., Verma, M., Dhankhar, R., Kaushal, V., Atri, R., & Sabharwal, R. (2015). Revisiting the role of phycocyanin in current clinical practice. International Journal of Pharmaceutical Sciences and Research, 6, 4588-4600. http://dx.doi.org/10.13040/IJPSR.0975-8232.6(11).4588-00
- Adjali, A., Clarot, I., Chen, Z., Marchioni, E., & Boudier, A. (2021). Physicochemical degradation of phycocyanin and means to improve its stability: A short review. Journal of Pharmaceutical Analysis, 12(3), 406–414. https://doi.org/10.1016/j.jpha.2021.12.005[CrossRef] [PubMed]
- Papalia, T., Sidari, R., & Panuccio, M. R. (2019). Impact of Different Storage Methods on Bioactive Compounds in Arthrospira platensis Biomass. Molecules, 24(15), 2810. https://doi.org/10.3390/molecules24152810[CrossRef] [PubMed]
- Güroy, B., Karadal, O., Mantoğlu, S., & Cebeci, O. I. (2017). Effects of different drying methods on C-phycocyanin content of Spirulina platensis powder. Ege Journal of Fisheries and Aquatic Sciences, 34(2), 129–132. https://doi.org/10.12714/egejfas.2017.34.2.02[CrossRef]
- Stanic-Vucinic, D., Minic, S., Nikolic, M. R., & Velickovic, T. C. (2018). Spirulina phycobiliproteins as food components and complements. In InTech eBooks. https://doi.org/10.5772/intechopen.73791[CrossRef] [PubMed]
- Schmid, B., Navalho, S., Schulze, P. S. C., Van De Walle, S., Van Royen, G., Schüler, L. M., Maia, I. B., Bastos, C. R. V., Baune, M., Januschewski, E., Coelho, A., Pereira, H., Varela, J., Navalho, J., & Rodrigues, A. M. C. (2022). Drying microalgae using an industrial solar dryer: A biomass quality assessment. Foods, 11(13), 1873. https://doi.org/10.3390/foods11131873[CrossRef] [PubMed]
- Stramarkou, M., Papadaki, S., Kyriakopoulou, K., Tzovenis, I., Chronis, M., & Krokida, M. (2021). Comparative Analysis of Different Drying Techniques Based on the Qualitative Characteristics of Spirulina platensis Biomass. Journal of Aquatic Food Product Technology, 30(5), 498–516. https://doi.org/10.1080/10498850.2021.1900969[CrossRef]
- Luo, G., Liu, H., Yang, S., Sun, Z., Sun, L., & Wang, L. (2024). Manufacturing processes, additional nutritional value and versatile food applications of fresh microalgae Spirulina. Frontiers in Nutrition, 11, 1455553. https://doi.org/10.3389/fnut.2024.1455553[CrossRef] [PubMed]
- Silva, N. C., Freitas, L. V. D., Silva, T. C., Duarte, C. R., & Barrozo, M. a. S. (2023). Use of Refractance Window Drying as an Alternative Method for Processing the Microalga Spirulina platensis. Molecules, 28(2), 720. https://doi.org/10.3390/molecules28020720[CrossRef] [PubMed]
- Silva, N. C., Graton, I. S., Duarte, C. R., & Barrozo, M. a. S. (2023). Effects of Infrared and Microwave Radiation on the Bioactive Compounds of Microalga Spirulina platensis after Continuous and Intermittent Drying. Molecules, 28(16), 5963. https://doi.org/10.3390/molecules28165963[CrossRef] [PubMed]
- Nakagawa, K., Ritcharoen, W., Sri-Uam, P., Pavasant, P., & Adachi, S. (2016). Antioxidant properties of convective-air-dried Spirulina maxima: Evaluation of phycocyanin retention by a simple mathematical model of air-drying. Food and Bioproducts Processing, 100, 292–302. https://doi.org/10.1016/j.fbp.2016.07.014[CrossRef]
- Ughetti, A., D’Eusanio, V., Strani, L., Russo, A. L., & Roncaglia, F. (2024). Influence of drying and storage conditions on the volatile organic compounds profile of Spirulina platensis. Separations, 11(6), 180. https://doi.org/10.3390/separations11060180[CrossRef]
- Fernandes, R., Campos, J., Serra, M., Fidalgo, J., Almeida, H., Casas, A., Toubarro, D., & Barros, A. I. R. N. A. (2023). Exploring the benefits of phycocyanin: from spirulina cultivation to its widespread applications. Pharmaceuticals, 16(4), 592. https://doi.org/10.3390/ph16040592[CrossRef] [PubMed]
- Khatun, R., Noor, P., Akhter, N., Jahan, M. A. A., Hossain, M., & Munshi, J. L. (2007). Spirulina Culture in Bangladesh XI: Selection of a Culture Medium, Suitable for Culturing a Local Strain of Spirulina. Bangladesh Journal of Scientific and Industrial Research, 41(3-4), 227-234. https://doi.org/10.3329/bjsir.v41i3.293[CrossRef]
- Jahan, M. A. A., Noor, P., Akhter, N., & Nahar, L. (1994). Spirulina Culture in Bangladesh III: Development of a medium suitable for commercial cultivation of Spirulina in Bangladesh. Bangladesh Journal of Scientific and Industrial Research, 29(4), 147-150.
- Kim, S.-G., Choi, A., Ahn, C.-Y., Park, C.-S., Park, Y.-H., & Oh, H.-M. (2005). Harvesting of Spirulina platensis by cellular flotation and growth stage determination. Letters in Applied Microbiology, 40(3), 190–194. https://doi.org/10.1111/j.1472-765X.2005.01654.x[CrossRef] [PubMed]
- Mufidatun, A., Koerniawan, M. D., Siregar, U. J., Suwanti, L. T., Budiman, A., & Suyono, E. A. (2023). The Effect of pH on Contamination Reduction and Metabolite Contents in Mass Cultures of Spirulina (Arthrospira platensis Gomont). International Journal on Advanced Science Engineering and Information Technology, 13(1), 84–90. https://doi.org/10.18517/ijaseit.13.1.16582[CrossRef]
- Chaiklahan, R., Chirasuwan, N., Loha, V., Tia, S., & Bunnag, B. (2011). Separation and purification of phycocyanin from Spirulina sp. using a membrane process. Bioresource Technology, 102(14), 7159–7164. https://doi.org/10.1016/j.biortech.2011.04.067[CrossRef] [PubMed]
- Pan-Utai, W., Kahapana, W., & Iamtham, S. (2017). Extraction of C-phycocyanin from Arthrospira (Spirulina) and its thermal stability with citric acid. Journal of Applied Phycology, 30(1), 231–242. https://doi.org/10.1007/s10811-017-1155-x[CrossRef]
- Boussiba, S., & Richmond, A. E. (1979). Isolation and characterization of phycocyanins from the blue-green alga Spirulina platensis. Archives of Microbiology, 120(2), 155–159. https://doi.org/10.1007/bf00409102[CrossRef]
- Bennett, A., & Bogorad, L. (1973). Complementary chromatic adaptation in a filamentous blue-green alga. The Journal of Cell Biology, 58(2), 419–435. https://doi.org/10.1083/jcb.58.2.419[CrossRef] [PubMed]
- Lauceri, R., Bresciani, M., Lami, A., & Morabito, G. (2017). Chlorophyll a interference in phycocyanin and allophycocyanin spectrophotometric quantification. Journal of Limnology. https://doi.org/10.4081/jlimnol.2017.1691[CrossRef]
- Porra, R. J. (2002). The chequered history of the development and use of simultaneous equations for the accurate determination of chlorophylls a and b. Photosynthesis Research, 73, 149–156. https://doi.org/10.1023/A:1020470224740[CrossRef] [PubMed]
- Blois, M. S. (1958). Antioxidant determinations by the use of a stable free radical. Nature, 181(4617), 1199–1200. https://doi.org/10.1038/1811199a0[CrossRef]
- Choi, W., & Lee, H. (2018). Effect of Ultrasonic Extraction on Production and Structural Changes of C-Phycocyanin from Marine Spirulina maxima. International Journal of Molecular Sciences, 19(1), 220. https://doi.org/10.3390/ijms19010220[CrossRef] [PubMed]
- IBM Corp. (2024). IBM SPSS Statistics for Windows (Version 30.0) [Computer software]. USA, IBM Corporation.
- Alp, D., & Bulantekin, Ö. (2021). The microbiological quality of various foods dried by applying different drying methods: a review. European Food Research and Technology, 247(6), 1333–1343. https://doi.org/10.1007/s00217-021-03731-z[CrossRef] [PubMed]
- Chentir, I., Hamdi, M., Li, S., Doumandji, A., Markou, G., & Nasri, M. (2018). Stability, bio-functionality and bio-activity of crude phycocyanin from a two-phase cultured Saharian Arthrospira sp. strain. Algal Research, 35, 395–406. https://doi.org/10.1016/j.algal.2018.09.013[CrossRef]
- Fratelli, C., Bürck, M., Silva-Neto, A. F., Oyama, L. M., De Rosso, V. V., & Braga, A. R. C. (2022). Green extraction process of food grade C-phycocyanin: biological effects and metabolic study in mice. Processes, 10(9), 1793. https://doi.org/10.3390/pr10091793[CrossRef]
- Katari, J. K., Khan, M. R. U. Z., Trivedi, V., & Das, D. (2023). Extraction, purification, characterization and bioactivity evaluation of high purity C-phycocyanin from Spirulina sp. NCIM 5143. Process Biochemistry, 130, 322–333. https://doi.org/10.1016/j.procbio.2023.04.022[CrossRef]
- Oliveira, E. G., Duarte, J. H., Moraes, K., Crexi, V. T., & Pinto, L. a. A. (2010). Optimisation of Spirulina platensis convective drying: evaluation of phycocyanin loss and lipid oxidation. International Journal of Food Science & Technology, 45(8), 1572–1578. https://doi.org/10.1111/j.1365-2621.2010.02299.x[CrossRef]
- Sobiechowska-Sasim, M., Stoń-Egiert, J., & Kosakowska, A. (2014). Quantitative analysis of extracted phycobilin pigments in cyanobacteria—an assessment of spectrophotometric and spectrofluorometric methods. Journal of Applied Phycology, 26(5), 2065–2074. https://doi.org/10.1007/s10811-014-0244-3[CrossRef] [PubMed]